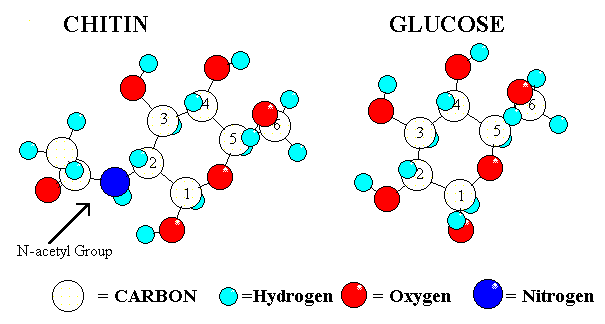
Although there has been used extencive resources on both research and development of processes and application, it's only the last two decades chitosan has been an industrial available product in large scale quantities. A polysaccharide. Chitosan is made by alkaline N-deacetylation of chitin, (Fig. 1). The term chitosan does not refer to a uniquely defined compound, it merely refers to a family of copolymers with various fractions of acetylated units. It consists of two types of monomers; chitin-monomers and chitosan-monomers.Chitin is a linear polysaccharide consisting of (1-4)-linked 2-acetamido-2-deoxy-b -D-glucopyranose. Chitosan is a linear polysaccharide consisting of (1-4)-linked 2-amino-2-deoxy-b -D-glucopyranose.Commercial, chitin and chitosan consists of both types of monomers.Chitosan is found in nature, to a lesser extent than chitin, in the cell walls of fungi.Chitin is believed to be the second most abundant biomaterial after cellulose. The annual biosynthesis of chitin has been estimated to 109 to 1011 tons. Chitin is widely distributed in the nature. Among several sources the exoskeleton of crustaceans consist of 15-20 % chitin of dry weight. Chitin found in nature is a renewable Bio-resource. Chitin is similar to cellulose both in chemical structure and in biological function as a structural polymer. The crystalline structure of chitin has been shown to be similar to cellulose in the arrangements of inter- and intrachain hydrogen bonding. It has been proposed to define chitosan and chitin as soluble or insoluble in 0.1 M acetic acid, respectively or by degree of deacetylation, >20 % of deacetylation is proposed defined as chitosan. Fig. 1. Structural formula of chitin and chitosan. Chitin
The main interest in chitosan derives from its cationic nature in acidic solutions, which provides unique properties relative to other polysaccharides which are usually neutral or negatively charged. The cationic nature allows chitosan to be used in among other applications waste-water treatment. Positively charged chitosans reacts with organic solids and cell surfaces which are usually negatively charged. Another useful property is that it is possible to control the solubility and the charge density by varying the mole fraction of N-acetylated units. Regarding potential applications of chitosan in water-solutions, control of the solubility of the chitosans is of primary importance. In general, the solubility of heteroglucans are influenced by the polarity and size of the monomers, distribution of the monomers along the chain, the flexibility of the chain, branching, charge density and molecular weight of the polymer. Product characterization. Both chitin and chitosan are naturally biodegrable and nontoxic products.Chitosan can be described in general by the following parameters:
All these parameters can be adjusted to the application chitosan is being used for. The main parameters that influence on the product properties is as mentioned above, the degree of deacetylation and viscosity. The deacetylation is very important to get a soluble product. But it`s not only the degree of deacetylation that influence on the solubility. The distribution of the acetyl groups are also important. Viscosity can be adjusted to each application, by controlling our process parameters. The range of viscosity is from 10 to 5000 cp. Applications. Chitosan is unique, with a polyamin character which makes it soluble (at acid pH), positively charged, different viscosity and easily modified chemically. These properties confer upon chitosan potential applications in:
For further information please contact : Dalwoo-chitoSan 3805-1 KWTC 159 Samsungdong kangnamgu Seoul, Korea Phone : 82 2 551 0938 Fax : 82 2 551 0940 Return to Dalwoo-chitoSan page.
Last updated April 12, 1999 Maintained by Dalwoo Corporation |